Bronsted-Lowry Acid
A proton (H+) donor in a chemical reaction.
Example: HCl donates H+ to water: HCl+H2O->H3O++Cl-.

Read the notes, then try the practice. It adapts as you go.When you're ready.
Session Length
~17 min
Adaptive Checks
15 questions
Transfer Probes
8
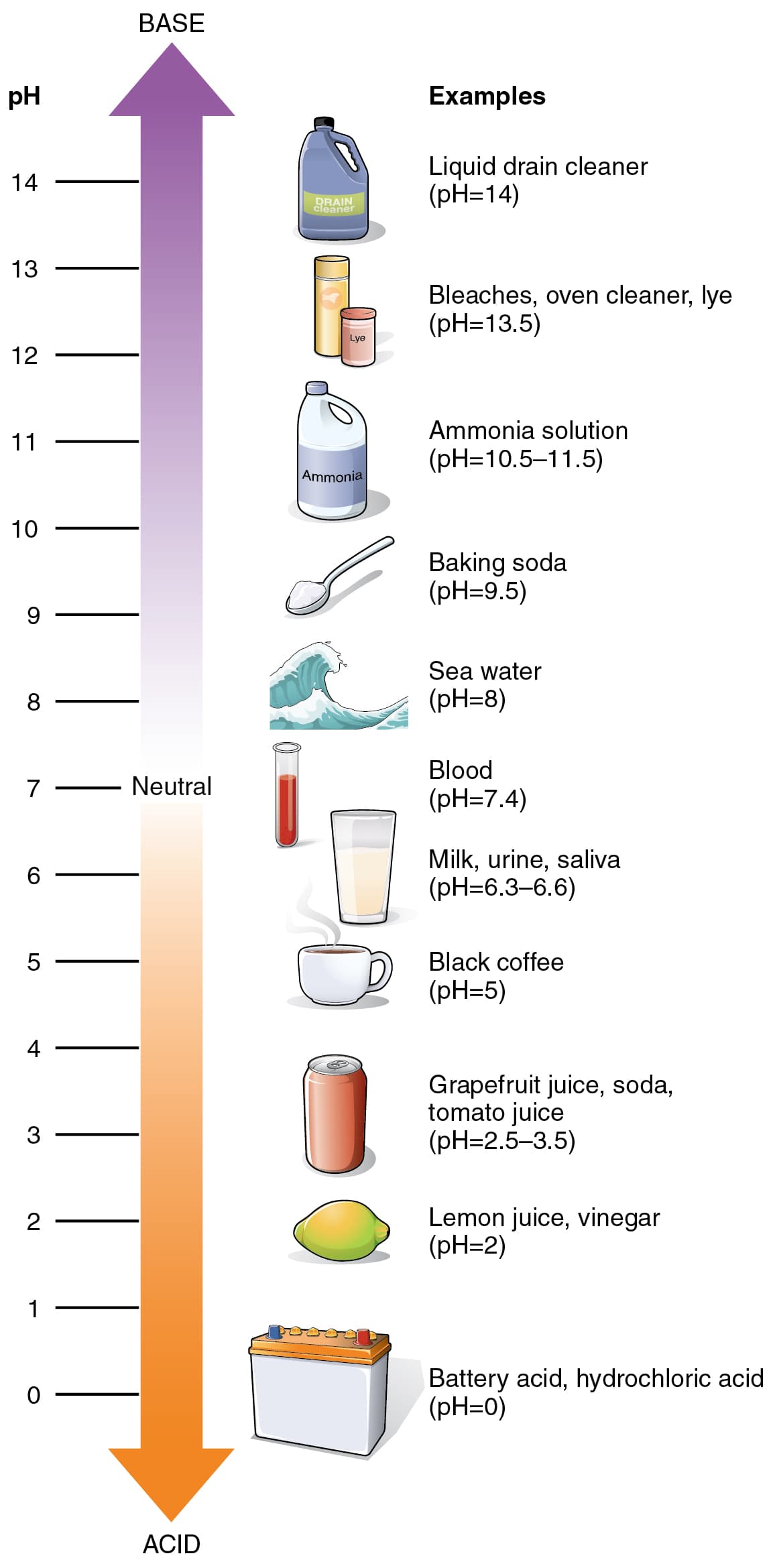
Acids and bases are central to chemistry, biology, and environmental science. This topic covers Arrhenius, Bronsted-Lowry, and Lewis definitions. Strong acids dissociate completely; weak acids establish equilibrium described by Ka.
pH quantifies acidity on a logarithmic scale. Buffers resist pH changes using conjugate acid-base pairs and the Henderson-Hasselbalch equation. Titration curves reveal equivalence points and indicator selection.
Covers pH calculations, Ka/Kb, buffers, titrations, polyprotic acids, and Lewis acid-base theory for AP Chemistry Unit 8.
One step at a time.
Adjust the controls and watch the concepts respond in real time.
A proton (H+) donor in a chemical reaction.
Example: HCl donates H+ to water: HCl+H2O->H3O++Cl-.
A proton (H+) acceptor in a chemical reaction.
Example: NH3 accepts H+: NH3+H2O<=>NH4++OH-.
Negative log of hydrogen ion concentration: pH=-log[H+].
Example: [H+]=1e-3 M gives pH=3.
Equilibrium constant for weak acid dissociation: Ka=[H+][A-]/[HA].
Example: Acetic acid Ka=1.8e-5.
pH=pKa+log([A-]/[HA]) for buffer solutions.
Example: Equal [A-] and [HA]: pH=pKa.
Solution resisting pH change, containing weak acid and its conjugate base.
Example: CH3COOH/CH3COO- buffer near pH 4.74.
Plot of pH vs volume of titrant added, showing equivalence point.
Example: Strong acid + strong base: equivalence at pH 7.
Electron pair acceptor.
Example: BF3 accepts electron pair from NH3.
Choose a different way to engage with this topic — no grading, just richer thinking.
Explore your way — choose one:
See how the key ideas connect. Nodes color in as you practice.
Walk through a solved problem step-by-step. Try predicting each step before revealing it.
This is guided practice, not just a quiz. Hints and pacing adjust in real time.
Small steps add up.
What you get while practicing:
The best way to know if you understand something: explain it in your own words.
More ways to strengthen what you just learned.